Environmental protection is becoming more and more pressing as our global society develops, and many countries in the world have issued regulations controlling the pollution caused by volatile organic compounds (VOCs), heavy metals, and other pollutants contained in paint.1
Such regulations and environmental considerations have boosted the development of environmentally friendly paints. As one paint of great importance in industrial applications, coil coating has seen an increasing demand causing the environmental requirements for such kinds of coatings to become more and more critical. Therefore, environmentally friendly coil coating will be the direction.
Coil coating is still the most efficient, economical, and environmentally sensitive means of applying paint to large areas of metal.2-3 The environmental and sustainability problems currently faced worldwide also call for the implementation and usage of renewable materials across industries, and the coil coatings industry in Europe has already moved to eliminate heavy metals, such as lead (Pb) and chromium (Cr), from formulations and has been using primers free of Cr(VI)4 for >10 years on some lines.
In the work presented here, an environmentally friendly coil coating primer was prepared, consisting of waterborne saturated polyester, waterborne crosslinking agents, and Cr-Pb-free pigments and fillers. The effects of pretreatments, crosslinking agents, pigments and fillers, and additives on the primer resistance and mechanical properties have been optimized and are discussed. The obtained crosslinked environmentally friendly coil coating primer exhibits excellent corrosion resistance and mechanical properties that were found to be comparable to those obtained on industrial reference oil-based primers.
Experimental Procedure
Materials
Water-dispersible acrylate-modified saturated polyester was used as binder resin. ZnMoO4, Zn3(PO4)2, Zn3Al(PO4)3, and AlH2P3O10 were selected as pigments, and powders of nano-titanium dioxide (TiO2), lithopone, and mica were selected as fillers. Commercialized waterborne blocked isocyanate DAP3257† and waterborne amino resins HD5717† and HD5735† were used as crosslinking resins. A small amount of waterborne dispersing agent, wetting agent, and leveling agent were also added.
Coil Coating Primer Preparation
First, the selected waterborne polyester, pigments, and deionized (DI) water were proportionally added to a small grinder and were reduced to a slurry with fineness <40 µm and good dispersion. Then, the crosslinking resins and additives were added to the slurry, followed by stirring at high speed for 2 h, filtration, and packaging for use. Galvanized steel sheets of 10 by 5 by 0.5 mm were pretreated by ultrasonic degreasing, rinsing, activation, drying, low-Cr(III) passivation, and again drying. The steel surface wet paint film was obtained using a scraper roller, and then oven baked at 225 °C for 40 to 45 s. The obtained coil coating samples were kept in a chamber with constant temperature and humidity for test and characterization.
Coil Coating Primer Characterization
A salt spray test (3.5 wt% sodium chloride [NaCl] solution) and a methyl ethyl ketone (MEK) scrubbing test were used to evaluate the primer corrosion resistance. In addition, a T-bend test, scratch test, and 2B pencil were employed to characterize the flexibility, adhesion, and hardness of the coil primer on galvanized steel samples, respectively.
Results and Discussion
Effect of Pretreatments on the Coating Primer Properties
In traditional coating systems, a primer is always applied to ensure adhesion between the pretreated steel surface and the topcoat.5 Pretreatment plays a key role in obtaining a qualified coil coating, and the usually employed pretreatment includes degreasing in alkaline solution → rinsing with water → surface conditioning → chemical conversion coating treatment → drying.6 To investigate the effect of pretreatment on the coating primer property, different pretreatments (degreasing; degreasing + low Cr(III) passivation; degreasing + activation + low-Cr(III) passivation) have been compared on galvanized steel sheets under the same controlled coating preparation conditions.
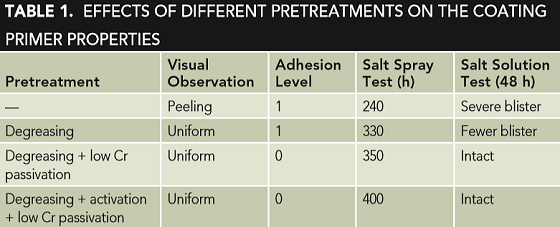
The obtained result is described in Table 1, and it shows that the pretreatment also has a crucial effect on the prepared waterborne coil coating primer. Without any pretreatment, Table 1 shows that primer peeling was observed, and the greases and/or rust present on the galvanized steel surface may lead to the poor primer adhesion.7 Both the adhesion and salt spray tests showed poor performance of the sample, and severe blistering was observed after the salt solution immersion test.
Compared with the sample of only pretreatment of degreasing, the samples of pretreatment of low-Cr(III) passivation exhibited good adhesion property and better corrosion resistance. This fact illustrates that low-Cr(III) passivation can greatly promote the mechanical properties and corrosion resistance of the primer due to the formed passive film.8
Moreover, it seems that the activation process in 3.5 wt% nitric acid (HNO3) solution also has an obvious beneficial effect in improving the primer properties as Table 1 shows, indicating that the presence of an unstable oxide film on galvanized steel surface is harmful for the primer quality.9-10 Therefore, in the present study, pretreatment of degreasing in alkaline solution → rinsing with water → activation in HNO3 → lowCr(III) passivation → drying was employed.
Effect of Pigments on Salt Spray Resistance of the Coating Primer
To highlight environmental friendliness, low-toxicity compounds of ZnMoO4, Zn3(PO4)2, AlH2P3O10, and Zn3Al(PO4)3 were selected as substitutes of chromates and lead pigments, and an appropriate amount of powders of nano-TiO2, lithopone, and mica were also added as fillers in consideration of the primer cost. The anticorrosion performance of selected pigments was evaluated by salt spray test. The pigment and additive contents used in the formulations during the test are expressed in weight (wt%) to the blinder resin, with a ratio of 1.3:1, and are listed in Table 2 along with the obtained results.
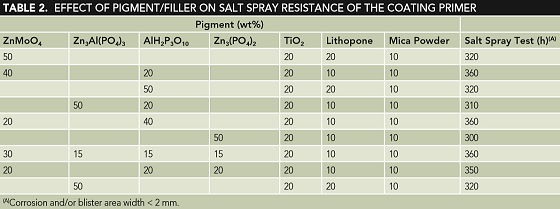
Table 2 shows that ZnMoO4, Zn3Al(PO4)3, and AlH2P3O10 have almost the same corrosion resistance, but Zn3(PO4)2 has a relatively lower corrosion resistance among the four selected pigments. In addition, the combined addition of the pigments can enhance the primer corrosion resistance to the salt spray test by >40 h. It was reported that MoO42– can adsorb on a galvanized steel surface with the formation of a passive film and passivate the steel.11 PO43– in Zn3Al(PO4)3 and Zn3(PO4)2 can form a complex protective film and make the oxidized substrate stable, and AlH2P3O10 also has the ability to stabilize the galvanized steel zinc oxide substrate and prohibit the anodic process.12 Therefore, the combined beneficial effect offered by the combined addition of the pigments improves the primer corrosion resistance as indicated by longer resistance to the salt spray test in Table 2. Taking consideration of cost and anticorrosion performance, in this investigation, pigments and fillers are used as follows (each to total pigments and fillers in wt%/wt%): 30% ZnMoO4, 15% Zn3Al(PO4)3, 15% AlH2P3O10, 15% Zn3(PO4)2, 20% TiO2 powder, 10% lithopone powder, and 10% mica powder.
Effect of Crosslinking Resin on the Coating Primer Properties
The usually employed crosslinking resins in coil coating are amino resin and waterborne blocked isocyanate.13 In consideration of the availability and compatibility with a waterborne polyester resin system, commercialized waterborne blocked isocyanate DAP3257 and waterborne amino resins HD5717 and HD5735 were selected for investigation, and other compositions in the primer were always kept the same. During the test, the crosslinking resin/binder resin ratio stays at 1:4, and the obtained effects of crosslinking resin on primer properties are listed in Table 3.
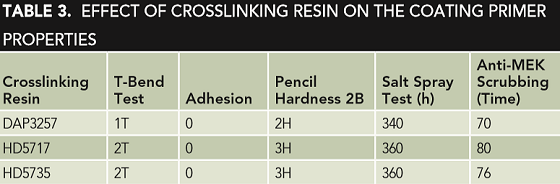
Table 3 shows that the effect of the three selected crosslinking resins on the primer properties is not obvious. In general, with blocked isocyanate being used, the primer film has a fine flexibility; with amino resin being used, the primer film has a higher brittleness and hardness. As to the anticorrosion performance, amino resins behave better than blocked isocyanate. It was reported that amino in amino resin can react with hydroxyl in polyester and the product has a much higher bond energy in a waterborne coating system; however, the formed urethane bond by reaction between isocyanate and polyester is not stable in water solution.14
In addition, HD5717 has a higher crosslinking degree than HD5735, and thus, it makes that primer coating provide better anti-MEK scrubbing resistance. Moreover, HD5735 is one type of all methyl etherified amino resins but HD5717 is one type of partial-methyl etherified amino resin, and thus, it needs a higher temperature or longer time to bake HD5735 resin to achieve the desired crosslinking degree. Based on the investigation, HD5717 amino resin was selected as the crosslinking resin for the coil coating primer.
Discussion of Additives on the Coating Primer Properties
As the selected binder resin has excellent shear stability, it can directly participate in the grinding of pigment slurry, and the resin structure is helpful for the wetting of pigments and fillers. Therefore, only a small amount of wetting dispersant is needed in the grinding process.
Compared with solvent-borne coatings, waterborne coatings have lower corrosion resistance. In addition, both the large surface energy of water and nonuniform evaporation of water during the baking process can add difficulties to both quality control and application of the waterborne coatings. Additionally, the surface performance of the coating is usually poor with the usual presence of an orange peel-like surface, small holes, wrinkling, and other defects, and thus, the coating surface is not sufficiently smooth with a poor leveling property.15
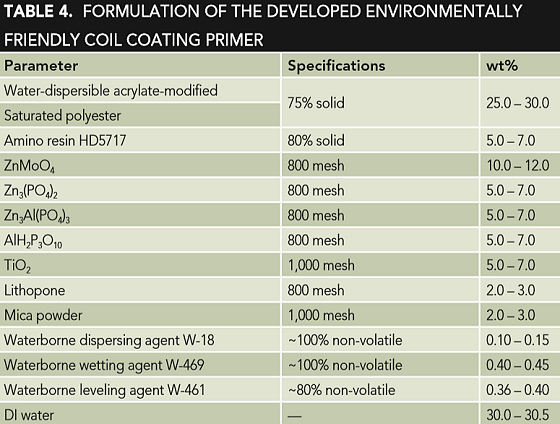
Therefore, to overcome these disadvantages as much as possible, a certain amount of water-based surface wetting agent and leveling agent were usually added to the waterborne coatings in practice, and excellent performance was achieved. Table 4 shows the formulation of an environmentally friendly coil coating primer developed in this investigation. When the obtained coating primer has a film thickness of 6 to 9 µm, it can have properties of resistance to 80 times MEK wiping, 2H pencil hardness, 0 grade adhesion, 0 grade T-bend, no change after 72 h immersion in 3.5 wt% NaCl solution, good resistance to sagging and leveling, and the salt spray resistance can reach 360 h.
Conclusions
An environmentally friendly polyester coil coating primer with low VOCs and no heavy metal contents has been developed by selection and evaluation of pigments and fillers, crosslinking resins, additives, and other main parameters. The developed waterborne coil coating primer has a salt spray test resistance of 360 h and ideal mechanical properties of 2H pencil hardness, 0 grade adhesion, and 0 grade T-bend. However, further improving the film surface properties and corrosion resistance still deserves our effort in future work.
† Trade name.
References
1 V.D. Athawale, R.V. Nimbalkar, “Waterborne Coatings Based on Renewable Oil Resources: an Overview,” J. of the American Oil Chemists Society 88, 2 (2011): pp. 159-185.
2 P.A. Sorensen, et al., “Anticorrosive Coatings: a Review,” J. of Coatings Technology and Research 6, 2 (2009): pp. 135-176.
3 M.J. Yang, J.D. Zhang, “Design and Cross-Linking Reaction of Blend of Reactive Polymer Latex Particles,” J. Mater. Sci. 40, 16 (2005): pp. 4,403-4,405.
4 M.C. Deya, et al., “The Influence of the Anion Type on the Anticorrosive Behaviour of Inorganic Phosphates,” Surface and Coatings Technology 150, 2-3 (2002): pp. 133-142.
5 S.W. Strunz, “Dielectric Relaxation in Barrier Coatings: A Square Root of Time,” Progress in Organic Coatings 39, 1 (2000): pp. 49-60.
6 W.S. Tait, “Using Electrochemical Measurements to Estimate Coating and Polymer Film Durability and Polymer Film Durability,” J. Coatings Technol. 75 (2003): pp. 45-50.
7 G. Bierwagen, et al., “EIS Studies of Coated Metals in Accelerated Exposure,” Prog. Org. Coat. 46, 2 (2003): pp. 149-158.
8 S.O. Han, L.T. Drzal, “Water Absorption Effects on Hydrophilic Polymer Matrix of Carboxyl Functionalized Glucose Resin and Epoxy Resin,” European Polymer J. 39, 9 (2003): pp. 1,791-1,799.
9 K. Ueda, H. Kanai, T. Amari, “Formability of Polyester/Melamine Pre-Painted Steel Sheets From Rheological Aspect,” Progress in Organic Coatings 45, 2-3 (2002): pp. 267-272.
10 N. Siyab, et al., “Going Green: Making Reality Match Ambition for Sustainable Coil Coatings,” J. of Coatings Technology and Research 13, 4 (2016): pp. 629-643.
11 A. Caretto, et al., “Fully Biobased Polyesters Based on an Isosorbide Monomer for Coil Coating Applications,” ACS Sustainable Chemistry and Engineering 6, 11 (2018): pp. 14,125-14,134.
12 T. Kolås, et al., “Cool Coatings With High Near Infrared Transmittance for Coil Coated Aluminium,” Solar Energy Materials and Solar Cells 196 (2019): pp. 94-104.
13 S. Gach, S. Olschok, U. Reisgen, “Geometric Modulation of Microscale Surface Structures for Defined Stimulation of Osteogenic Differentiation on Implant Materials,” Materialwissenschaft und Werkstofftechnik 50, 4 (2019): pp. 365-371.
S.Y. Liu, et al., “High-performance Cu/Al Laminated Composites Fabricated by Horizontal Twin-Roll Casting,” Materialwissenschaft und Werkstofftechnik 49, 10 (2018): pp. 1,213-1,223.
15 J.D. Zhang, et al., “Synthesis and Characterization of Crosslinkable Latex with Interpenetrating Network Structure Based on Polystyrene and Polyacrylate,” Polymer International 55, 8 (2006): pp. 951-960.