Editor’s note: Learn more about reinforced concrete corrosion in this Materials Performance quarterly special feature, “The Science Behind It.” After you've read the MP article on verifying the validity of silane as an additive by monitoring the corrosion behavior of silane-enhanced mortar, explore the science behind the corrosion problem, which is presented in several related CORROSION articles listed at the end of the article.
Concrete is often exposed to aggressive environments. Due to its porous structure, aggressive agents (e.g., water, carbon dioxide [CO2], and chloride and sulfate ions) in the environment have caused the deterioration of concrete, resulting in the decrease of its service life.1 A variety of methods have been applied that upgrade the durability of concrete structures by reducing the concrete’s permeability. The application of polymer coatings is a technique that reduces the ingress of moisture and ions.2 One of the methods used to protect the concrete surface from corrosion caused by moisture is hydrophobization, and silanes are one of the common surface hydrophobization treatment agents for concrete structures.3
Although research has shown that a silane surface treatment and siloxane polymers seem promising for natural aggregate concrete, fewer studies have been conducted to investigate polymer concrete using silane as an integral additive for the improvement of the durability of mortar and concrete.4 Therefore, the present work is intended to verify the validity of silane as an additive by monitoring the corrosion behavior of silane-enhanced mortar. Silane coupling agents were used in varying proportions, and the effect of the coupling agent content on mortar properties was studied.
Experimental Procedures

Materials and Preparation
The cement used was No. 42.5 ordinary Portland cement (OPC) made in China. The integral silane mortar additive was (γglycidoxypropyl) methyldimethoxysilane (KH560). River sand with a fineness modulus of 2.43 was used as the fine aggregate. A water/cement ratio of 0.4 was used for preparing mortar mixtures incorporating no silane (the reference sample), and 1, 3, and 5% silane. Distilled water was used to prepare the mortars.
Imperviousness Test
The imperviousness test was conducted according to the Chinese Standard JGJ/T 702009.5 All measurements were made at a temperature of 20 ±2 °C, with relative humidity varying from 70 to 80%. The results presented are the average of three measurements.
Crack Resistance
Crack resistance of modified and unmodified mortar was determined in accordance with the flat test suggested by ACI Committee 544.6
Carbonate Resistance
Carbonation depth was measured in an accelerated carbonation experiment. The two ends of each specimen were sealed with epoxy resin and stored in a carbonation chamber with 4% CO2 by volume.
Freeze-Thaw Resistance
Samples were tested in a freeze-thaw testing machine for 50, 100, 150, and 200 cycles with a temperature range of –25 to 20 °C. After the freeze-thaw test, the weight loss rate and compressive strength loss rate of the samples were determined using Equation (1):
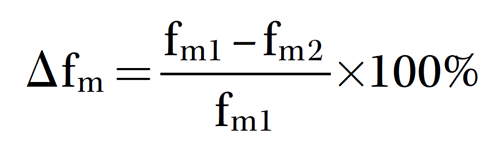
where Δfm equals the compressive strength loss rate after the freeze-thaw test (%), fm1 equals the compressive strength (MPa) of the samples before the freeze-thaw test, and fm2 equals the compressive strength (MPa) of the samples after the freeze-thaw test.
Scanning Electron Microscopy
The morphology of the mortar samples was investigated using scanning electron microscopy (SEM). The samples were investigated via SEM conducted on an FEI Sirion 200† field-emission scanning microscope at an acceleration voltage of 25 kV.
Results and Discussion
Imperviousness Test
The imperviousness test results are shown in Figure 1. The imperviousness pressure was 2.0 MPa for the reference sample. The imperviousness pressure of the mortar mixture prepared with 1% silane was 2.2 MPa, which was 10% more than the corresponding mortar reference sample. Furthermore, the imperviousness pressure values for mortar samples with 3 and 5% silane were 2.45 and 2.8 MPa, respectively. Therefore, the silane additives were very effective in improving the mortar’s imperviousness. The hydrophobic nature of silane and coverage of microvoids in the mortar were mainly responsible for the sharply enhanced imperviousness.2
Crack Resistance
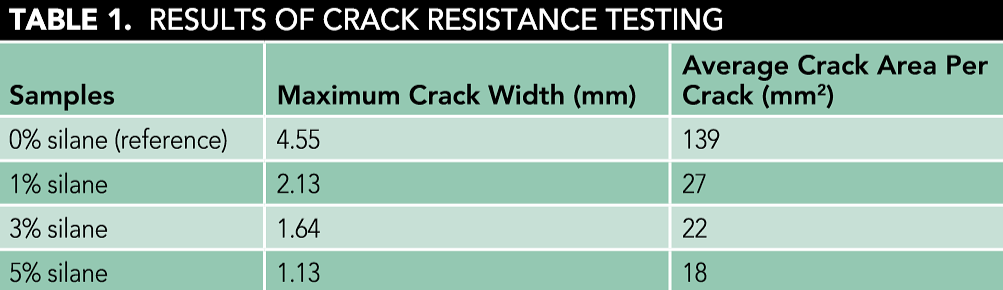
The results of the mortar’s crack resistance with different silane dosages are shown in Table 1. For the reference sample, the maximum crack width and crack area per crack were 4.55 mm and 139 mm2, respectively. The addition of silane reduced the maximum crack width and crack area per crack. Compared to the reference sample, the maximum crack width and crack area of the sample with 1% silane were greatly reduced—to 2.13 mm and 27 mm2, respectively. Further increases in the silane dosage also decreased the maximum crack width and crack area per crack. The silane significantly improved the crack resistance of the mortar samples. This can be explained by the fact that silane hydrolyzed and the functional group 4-SiOH was obtained.7 The reactive silanol groups became anchored to the cementitious materials or to the aggregate and reinforced the interface between the silane and mortar. Because of the bridging action of the silane, the crack resistance of mortar was improved.

The results presented in Figure 2 compare the waterproofing properties of unmodified (left) mortar and mortar with 5% silane (right) when the cracks appear. This comparison demonstrates that the mortar samples with silane showed excellent waterproofing properties even after the cracks occurred.
Carbonation Resistance
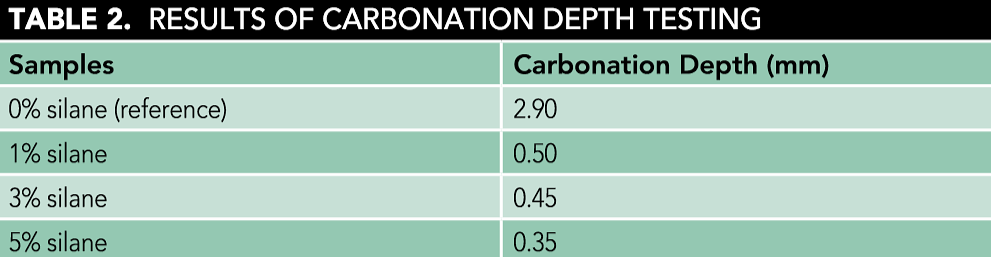
Table 2 shows the relationship between silane dosage and carbonation depth at 3, 7, 14, and 28 days of carbonation. For mortar samples with silane, the carbonation depths were lower when compared to the reference sample. Furthermore, as the silane dosage increased, the carbonation depth consistently decreased. This can be ascribed to SiOH groups that are produced by the hydroxylation of silane.8 Because of the self-polycondensation of SiOH groups, the siloxane polymer network can be obtained. Thus, the carbonation resistance of mortar was improved due to the inherent advantages of siloxane, including excellent thermal stability, good water rejection characteristics, and resistance to carbonation and chemicals.
Freeze-Thaw Cycles
Figure 3 shows the loss ratio of samples at 50, 100, 150, and 200 cycles for the mortar reference sample and samples with different silane additives. The mortar containing silane additives exhibited lower loss of compressive strength compared to that of the reference mortar, indicating that the mortar samples show better freeze-thaw resistance after the silane addition. This can be explained by reason of the “coupling” effect of the silane. In addition, the hydrophobic nature of silane and the coverage of microvoids also reduced the moisture permeability of mortar and enhanced the freeze-thaw resistance.
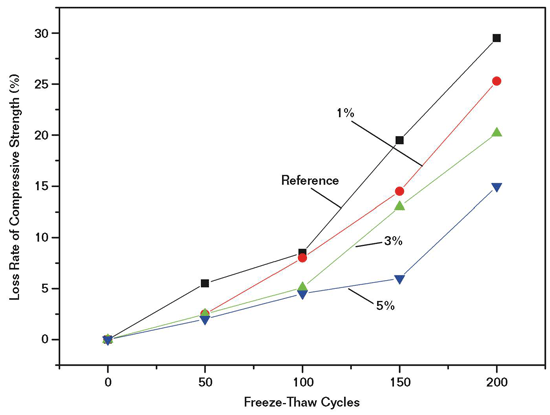
Figure 4 shows the microstructures of the reference mortar and mortars with silane. The SEM image of the reference mortar shows that an abundance of hydrated phases—calcium hydroxide [Ca(OH)2], CH, and calcium silicate hydrate (CSH)—exist in the sample. For the mortars with 1% silanes, the silanes encase the mortar particles firmly. Further increases in the silane dosage results in the denser network of silane polymers. These findings help to explain the improvement of the mortar durability after the silane was added. Silanes perform a networking effect that holds mortar particles together, which improves the freeze-thaw behavior and carbonation corrosion resistance, as well as the imperviousness and cracking resistance.

Conclusions
The objective of the present study was to evaluate the effects of silane on the durability of mortar samples. Thus, laboratory tests were conducted to evaluate the durability of mortar containing different silane dosages.
• The silane additives were very effective in improving the mortar’s imperviousness and freeze-thaw resistance due to the hydrophobic nature of silane.
• Because of the bridging action of the silane, the crack resistance of mortar was improved.
• The carbonation resistance of mortar was improved due to the inherent advantages of siloxane, such as carbonation and chemical resistance.
• The SEM images revealed that the siloxane polymer network improved the durability of mortar.
†Trade name.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (51409203). The authors express their great appreciation of the funding supported by Research and Innovation Initiatives of WHPU (2015d3) and State Key Laboratory of Silicate Materials for Architectures (Wuhan University of Technology SYSJJ201613).
References
1 H. Shan, et al., “A Novel Electrochemical Technique for Enhancing Silane Penetration Depth into Mortar,” Constr. Build. Material 144 (2017): pp. 645-649.
2 Z. Liu, W. Hansen, “Effect of Hydrophobic Surface Treatment on Freeze-Thaw Durability of Concrete,” Cement Concrete Comp. 69 (2016): pp. 49-60.
3 D. Barnat-Hunek, P. Smarzewski, “Influence of Hydrophobisation on Surface Free Energy of Hybrid Fiber Reinforced Ultra-High Performance Concrete,” Constr. Build. Material 102 (2016): pp. 367-377.
4 L. Jiesheng, et al., “Properties of Polymer-Modified Mortar Using Silane as an Integral Additive,” J. Test. Eval. 1 (2016): pp. 175-182.
5 JGJ/T 702009, “Standard for Test Method of Basic Properties of Construction Mortar” (Beijing, China: MOHURD, 2009).
6 “Report on the Physical Properties and Durability of Fiber-Reinforced Concrete,” American Concrete Institute, ACI 544.5R10, March 2010.
7 C. Zhou, et al., “Corrosion Resistance of Novel Silane-Functional Polybenzoxazine Coating on Steel,” Corros. Sci. 70 (2013): pp. 145-151.
8 L. Jiesheng, W. Shaopeng, E. Dong, “Effect of Coupling Agent as Integral Blend Additive on Silicone Rubber Sealant,” J. Appl. Polym. Sci. 4 (2013): pp. 2,337-2,343.