For the foreseeable future, fossil fuels will continue to be the dominant source of the world’s primary energy production. There has been growing concern that the use of these carbon-based fuels produces greenhouse gases, principally carbon dioxide (CO2), which adversely affects the global climate and environment. One way to mitigate the problem is to use carbon capture, transportation, and storage (CCTS) techniques and systems. The CCTS process involves capturing CO2 from the atmosphere, compressing the gas, and transporting it to a long-term storage site. Transmission may be accomplished by pipelines, tankers, trains, trucks, compressed gas cylinders, as CO2 hydrate, or as solid dry ice. However, only pipeline and tanker transmission are reasonable options for the large quantities of CO2 associated with power stations, hydrocarbon production, and other industrial activities.
“CCTS may be considered a short- to medium-term mitigation avenue to reduce greenhouse emissions and allow sufficient carbon-neutral technological advances to mature,” says NACE International member and Fellow Bijan Kermani, editor of the new NACE book, Carbon Capture, Transportation, and Storage—Aspects of Corrosion and Materials. “Until these new technologies are developed and applied, corrosion engineers are focusing on how to best design pipelines for CO2 transport through materials selection and other means.”
Kermani invited four leading experts on the topic of CCTS to answer a series of questions on the challenges involved with preventing pipeline corrosion of these CO2 transmission systems. NACE International Fellows Sankara Papavinasam, Arne Dugstad, and Srdjan Nešic, and NACE member Yoon-Seok Choi provided the following comments on pipeline corrosion issues related to CCTS.
Q: What corrosion mechanisms and challenges are involved with transporting and storing CO2?
Sankara Papavinasam: Carbon steel (CS) is the primary material for constructing CO2 transportation pipelines. CO2 dissolves in water to form carbonic acid, which is corrosive to CS. The rate of corrosion or weight loss depends on the formation and stability of iron carbonate that is formed as a consequence of corrosion of CS in carbonic solution. Corrosion of CS in aqueous CO2 has been an active field of study for the last 50 years. The corrosion rates reported in the studies can vary by four to six orders of magnitude. These variations may be due to several factors including the use of different laboratory methodologies, the presence of other chemicals, various operating conditions (i.e., temperature, flow, composition, and pressure), variation in the compositions of CS, duration of experiments, and lack of care in conducting the experiments.
Recently many groups have published results on corrosion rates under CCTS conditions. The salient features are summarized as follows:
• Interaction between the material and the environment is not an issue, even in the presence of several impurities as long as free water is not present.
• With current knowledge and experience, transportation and injection into a well can be safely carried out; however, material-environmental issues with different capture systems and long-term behavior under storage conditions are not well understood.
• The absence of standard methodology to investigate the CCTS issues has produced research and development results that may be of very limited use.
• Similar to the corrosion rates of materials in an aqueous CO2 environment, the corrosion rates of materials in CO2 sequestration environments vary considerably. Even though all studies indicate that the corrosion rate decreases with time, in general abnormally high corrosion rates have been reported.
• It appears that the lack of coordinated and collaborative research efforts continues to produce corrosion rates in the laboratory that cannot be meaningfully used to select materials for a CCTS network.
Arne Dugstad: If an aqueous phase is formed, a number of competing corrosion mechanisms can be foreseen depending on the type of impurities that are dissolved in the water phase and the type of cross-chemical reactions (bulk phase reactions) taking place. Experiments have shown that an aqueous phase containing elemental sulfur and sulfuric and nitric acid in addition to O2 and H2S can form when the CO2 stream contains water, NO2, SO2, H2S, and O2 in concentrations well within the limits suggested in many of the published recommendations for maximum impurity concentrations in CO2. 1
In order to predict and mitigate corrosion and solids (elemental sulfur and corrosion products) formation, there is a strong need to better understand the relationship between the water content and the maximum concentration of other impurities that can be present before separate corrosive phases are formed or cross-chemical reactions take place. At present, there is a lack of data and therefore it is not possible to define the limits for the various impurities when they are mixed.
Srdjan Nešic and Yoon-Seok Choi: The corrosion mechanism involved for transporting and storing CO2 is similar to the CO2 mechanism in oil and gas applications. However, particular conditions involving the source of CO2; for example, flue gas, can introduce more complicated mechanisms such as the presence of contaminants from products of combustion.
Q: Why are pipelines one of the best options for transporting CO2 associated with power stations, hydrocarbon production, and other industrial activities compared to other methods?
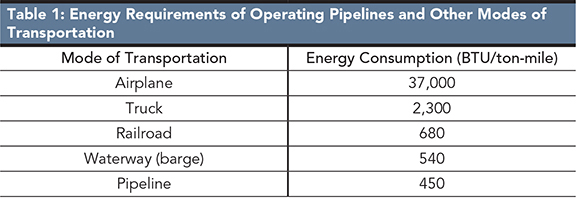
SP: Pipelines are the most convenient mode for transportation of large quantities of crude oil and gas. Advantages include lower cost compared to other modes of transportation (Figure 1)2, the energy required to operate pipelines is lower than that required to operate other modes of transportation, and overall they are environmentally friendly, safe, reliable, convenient, and less sensitive to inflation when compared to other modes of transportation (Table 1)2. The practice of transporting liquid CO2 through pipelines for oil recovery has been in practice for several years.

AD: Following the “Blue Map Scenario”3 for the abatement of climate change, about 10 Gtons of CO2 need to be safely transported and stored underground in 2050. This huge amount of CO2 requires the construction of about 3,000 12-in (0.3-m) (or 1,000 20-in [0.5-m]) pipelines assuming a flow velocity of 1.5 m/s. The only cost-effective material that can be used for such an extensive network is CS. Pipelines are regarded to be the best option in the long run for transporting CO2 to permanent storage, but ship transport is an alternative as it gives more flexibility, particularly in the early phase of CCTS and until pipelines are established for that purpose.
SN and Y-S C: CO2 pipelines are not new. Gaseous CO2 is typically compressed to a pressure above 8 MPa in order to avoid two-phase flow regimes and increase the density of the CO2, thereby making it easier and less costly to transport. In some situations or locations, transport of CO2 by ship may be economically more attractive, particularly when the CO2 has to be moved over large distances or overseas.
Q: What are the materials selection and optimization considerations for pipelines designed to transport CO2?
SP: CS is the ideal choice to construct a CCTS network. Most secondary oil recovery systems (for which CO2 is currently being transported) are constructed using CS when the amount of water and impurities can be strictly controlled. When these variables can’t be controlled, corrosion-resistant alloys (CRAs) are considered. Appropriate CRAs are selected based on laboratory testing and evaluation and the limits of their application.
AD: It is important to note that coated pipelines and use of soft materials need special attention due to swelling, blistering, and delamination issues during depressurization.
SN and Y-SC: Minimum standards for “pipeline quality” CO2 should emerge as the CO2 pipeline infrastructure develops further. It is unlikely to be practical to transport wet CO2 in low-alloy CS pipelines because of this high corrosion rate. If the CO2 cannot be dried, it may be necessary to build the pipeline of a CRA. However, the cost of steel has greatly increased recently and this may not be economical.
Q: Are there corrosion prediction models that can be applied to transportation systems for CO2 and if so, can you briefly describe them?
SP: Currently no reliable prediction models are available for CCTS conditions, but industry has been successfully transporting supercritical CO2 based on sound scientific and technical knowledge. In order to develop a reliable and uniformly acceptable model, researchers, and stakeholders should:
• Coordinate to write a position paper on key issues in understanding material-environmental interactions with respect to carbon sequestration networks (CCTS)
• Perform pre-round robin experiments to evaluate the various methodologies being used to conduct CCTS experiments
• Conduct round robin experiments based on the outcome of pre-round robin experiments using only promising laboratory methodologies and procedures
• Develop standardized methodologies for the evaluation of CCTS materials
To select appropriate materials for CO2 transmission pipelines, laboratory experiments must be conducted at supercritical conditions to investigate the influence of various impurities including O2, N2, SO2, NOx, H2S, ethylene glycol, amines, and ammonia. Before the influence of impurities can be investigated, the repeatability and reproducibility of the proposed laboratory methodology in the pure CO2 phase must be understood. Only models developed based on the above considerations should be accepted by stakeholders and industry.
AD: There are several corrosion prediction models that are used to assess CO2 corrosion rates in oil and gas environments. Some of these oil and gas models might be extended to high CO2 partial pressure and used under upset conditions with significant ingress of pure water or brine. To our knowledge there are no models that predict when corrosion takes place in a dense phase CO2-containing combination of impurities like SOx, NOx, H2S, O2, and water. Models are also required that predict formation of elemental sulfur and possibly other solid reaction products.
SN and Y-SC: There is one corrosion model available in public domain, described in the article, “A Mechanistic Model of Pipeline Steel Corrosion in Supercritical CO2-SO2-O2-H2O Environments.”4 It was established based on the CO2 corrosion models and the atmospheric corrosion model.
Q: What are the most effective corrosion mitigation methods for pipelines carrying large quantities of CO2 to facilities for long-term storage?
SP: The most effective method to mitigate corrosion of CCTS networks is to limit the amount of water. The presence of water in CO2 pipelines produces carbonic acid (which is corrosive) and hydrate (which blocks the flow). For this reason the water content is kept low and is continuously monitored. Typical water content allowing full dehydration is less than 10 lb of water per million standard cubic feet of CO2.
AD: It is critical to prevent formation/precipitation of separate aqueous phases. In other words, keep the combination of impurities within limits that do not give bulk phase reactions and form aqueous phases.
SN and Y-S C: Sufficient drying (water removal) upstream of the pipeline is required to prevent breaking out of free water and excessive corrosion rates, but it contributes to an increase in handling cost, especially for offshore installations where weight allowance and space for process equipment installation are very restricted. Using a CRA such as 13Cr steel can be an option; however, application of 13Cr steel is limited to milder environments (CO2/O2 environments) due to the low corrosion resistance in SO2-containing environments. A corrosion inhibitor would be the most effective corrosion mitigation methods for this application. However, one should consider the presence of impurities (SO2, O2, NO2, etc.) and the compatibility of inhibitors with liquid and supercritical CO2.
Q: What are some of the research and technology activities underway for CCTS, and what should future efforts involve?
SP: When CO2 capture infrastructure and pipelines accidentally release CO2, the CO2 will replace oxygen in the area, making it an unsafe zone. The magnitude of the areas affected depends on the pressure (which in turn to some extent depends on materials) and the magnitude of the leak. The applicability of currently available models to impurity-containing CO2 is being evaluated. A dense phase release model is being developed to establish procedures for evacuation to a safe distance from CO2 infrastructure.
It is known that CO2 may decompose sealing materials made from petroleum products when the pressure is reduced. Therefore, specially made sealants from inorganic materials and grease should be used. However, the material properties in dense phase CO2 are not well established. To overcome this issue, systematic research is being conducted to develop an open literature database on compatibility of various non-metals (plastics, ceramics, and polymers) in dense phase CO2 containing various impurities including but not limited to O2, N2, SO2, NOx, H2S, ethylene glycol, amines, and ammonia.
When a real gas expands from a region of higher pressure to a region of lower pressure, the temperature of the system decreases (Joule-Thomson effect). The effect of this phenomenon during the injection of CO2 underground has been investigated and found to be relatively minimal because of the porosity and permeability of the relevant rock formations. However, when CO2 is released from an operating CO2 infrastructure accidently, the resulting J-T effect will cool the structure in contact with the escaping CO2 and, if it is made of steel as is highly likely, can embrittle it. This can lead to fracture of the steel and the resulting cracks can run for long distances. It will be necessary to ensure that any cracks that are initiated will be arrested.
The problem is exacerbated by the fact that CO2 exists as a two-phase mixture over a large range of velocities during release, and the pressure at the crack tip is therefore maintained at a high level during crack propagation. This phenomenon is more pronounced for CO2 than for natural gas, so arrest of a running crack may be even more difficult for CO2 pipelines than for natural gas pipelines. To understand these issues, systematic research is being conducted to establish whether the J-T cooling phenomenon will cause embrittlement in the potential materials to be used in the CCTS network.
AD: These are topics that the Institute of Energy Technology focuses on and that need more attention:
• Field experience and most lab experiments show that dry pure CO2 and pure CO2 that contains dissolved water well below the saturation limit in the pure CO2-H2O system is noncorrosive to CS under pipeline transportation conditions, but what is the consequence of accidental ingress of a free water phase? The corrosion rates measured in lab experiments, with steel specimens fully submerged in a water phase in equilibrium with pure dense phase CO2, were in the range of 3 to 40 mm/y for temperatures between 4 to 50 °C. A strategy for handling accidental water ingress is required.
• If dry CO2 continues to flow after a water incident, it is assumed that the water will be dissolved quickly and not seriously threaten the integrity of the pipeline. Continuous water ingress or long-lasting shutdown after water ingress will give quite a different situation. At shutdown it might be necessary to remove the water in the pipeline. Water removal includes depressurization of the pipeline and experience from existing pipelines indicates that this can take weeks. The acceptable response time after water contamination will be system specific and depend on the corrosion rate and corrosion allowance. Presently, the corrosion rate in a pipeline suffering from accidental water ingress cannot be estimated accurately due to lack of corrosion data.
• There is very little published data on the effect of flue gas impurities like SOx, NOx, and O2. Experiments have shown that corrosion takes place at water concentrations far below the water solubility in pure water-CO2 systems when such impurities are present. Particularly large effects and high corrosion rates (>1 mm/y) are observed in the presence of NO2.
• There is a strong need to better understand the relationship between the water content and the impurity concentration at which corrosive phases form and corrosion takes place. At present, there is a lack of data and therefore it is not possible to define the safe limits for the various impurities when they are mixed.
• Depressurization of the pipeline will increase the concentration of many of the impurities (including water, SOx, and NOx) in the remaining liquid phase. In order to quantify the increased corrosivity after depressurization, there is a need for more solubility and partitioning data for the water-dense phase CO2 system.
• If glycol (i.e., MEG, DEG, TEG) and amine carryover form a separate phase, it will extract water and form a corrosive environment. If the CO2 is contaminated with glycols, the solubility and possibility of forming separate aqueous phases should be investigated.
• Amine and ammonia carried over from the CO2 capture process will dissolve in a free water phase. This will increase the pH while the potential corrosion rate (the corrosion rate without protective layers on the steel surface) might be reduced. The effect of pH adjustment or pH stabilization is not well documented and should be investigated in more detail.
• Solids in the pipeline can be hygroscopic and become wetted at water concentrations below the solubility limit for bulk phase precipitation. For instance, solid phases might be present in old pipeline systems that are converted to CO2 transportation lines.
• Corrosion prediction and solids formation models that cover CO2 pressures up to several hundred bar and take the true corrosion and reaction mechanisms into account will be needed in order to predict the corrosion rates in CCTS pipelines. And even more important, the models have to include the effect of impurities like SOx, NOx, and O2. None of the existing models can presently be used and an extensive amount of experimental corrosion and solubility data will be needed in order to develop a model.
• Little data on CRA corrosion are found in the literature. It is reported that 13 %Cr corroded at the same rate as CS when a reasonably large amount of SO2 was present. Attack on duplex steel has been observed in systems with small amounts of water, SO2, and NO2. More data are needed in order to define a safe operation window.
SN and Y-SC: We would focus on evaluating and selecting suitable corrosion inhibitors, and developing a mechanistic corrosion model.
Q: What are the impurity limits in terms of water, H2S, and other impurity gases that can be tolerated using CS lines?
SP: The CO2 being captured from the power plant may contain many impurities depending on the process and capture technologies. In general maximum impurity levels in transportation pipelines are specified; for example, in standards such as ASME B31.8.5 Typical compositions of CO2 pipelines are 98% CO2, 0.15 to 1.5% nitrogen, and 0.11 to 1.5% methane.
Another effect of impurities is to change physical properties such as critical pressure. Critical pressure is important because this pressure determines the state (gas or supercritical fluid) of the CO2. The impurities may have an impact on the operating regime of a pipeline. For example, it may have to be operated at a different pressure regime than one that would be required if pure CO2 were transported.
Impurities may also have an effect on the pressure and temperature drops. A larger pressure drop means an increased number of compressor stations to operate CO2 pipelines. From the safety perspective, a sudden drop in temperature will cause embrittlement of materials as well as hydrate formation, both of which could potentially damage the pipeline.
CO2 transported in the CCTS network may contain various impurities including but not limited to O2, N2, SO2, NOx, H2S, ethylene glycol, amines, and ammonia. While the level of water allowed is established, the limits of impurities and their influence on the materials behavior is not yet well established.
AD: Although dense phase CO2 has been transported for more than 30 years, there is limited knowledge about possible corrosion and bulk phase reactions when the CO2 contains flue gas impurities like SOx, NOx, O2, and CO, in addition to H2O and H2S. A number of CO2 specifications and recommendations for maximum impurity concentrations have been published. The most referred CO2 quality recommendation has been suggested in the DYNAMIS project.6 Other frequently referred CO2 specifications have been presented by IPCC7 and Kinder Morgan.8 The National Energy Technology Laboratory (NETL) recently issued a table of recommended limits for CO2 stream compositions based on a review of 43 CO2 specifications found in the literature.9 A large variation in the reported impurity concentrations can be seen and that is reasonable as the impurities in the CCTS/CCUS stream will depend on the fuel type, the energy conversion process (post-combustion, pre-combustion, or oxyfuel), and the capture process. In addition, with new capturing technologies, new compounds (impurities) can be formed and higher concentrations of impurities can follow the CO2 phase with an unknown effect on corrosion.
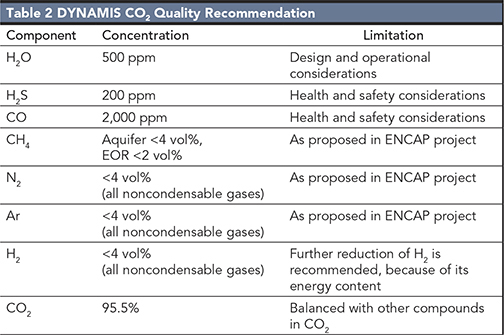
When pipeline design philosophy for CO2 transportation is discussed it is commonly accepted that the CO2 should be sufficiently dry to prevent drop-out of a separate aqueous phase in any part of the pipeline, as free water can give both corrosion and hydrate formation. There is, however, no consensus on what the actual target should be for the maximum water concentration. The main reason for the uncertainty is lack of field experience and laboratory data. It has been argued that extensive dehydration down to 50 ppmv should be applied. This limit has been specified for the first CO2 pipelines in the United States10 and for the Snøhvit11 pipeline in Norway. Other specifications are less conservative as suggested in the DYNAMIS project (500 ppmv) and for the Kinder Morgan pipeline (650 ppmv). All the suggested values are well below the water solubility given in pure dense phase CO2.
SN and Y-S C: There are several quality recommendations for CO2 pipelines. One example is from the DYNAMIS project (Table 2)6
Q: Do you have any other comments on pipeline corrosion and CO2 transport?
SP: With respect to CCTS network development, the technology is already available (in terms of transporting CO2 for oil recovery). The main issue is the magnitude of the amount to be transported. Unlike other fields in which the development of infrastructure is predominantly funded and developed by industry, the funding for a CCTS network would come predominately from the government. As a consequence, the activities and knowledge will be scrutinized more by stakeholders and the public. For this reason, it is the responsibility of corrosion professionals to clearly understand and articulate the real corrosion issues and develop solutions for them. Temptations to publish data from short-term tests and “reinventing the wheel type of research” should be avoided. Technical associations like NACE International should continue to take a leadership role to ensure that appropriate solutions (for which there currently are no solutions) are developed. Further, these associations should continue to disseminate relevant information in the form and language that is easily understood by the general public.
Bijan Kermani: In general, corrosion monitoring and inspection used for pipelines are applicable to pipelines transporting CO2. However, as corrosion damage may not follow normal trends, some specific innovative methods may be required to allow detection of impurities, water content, and related parameters. Inspection methods also need a review to determine their applicability to CO2 lines and whether special measures are required.
In closing, we need to develop and deliver processes that allow safe CCTS in the future. Great progress has and is being made to address individual elements of CCTS systems. However, there is a need to integrate the activities of all elements of CCTS, focusing on gap analysis and economic impacts as the first step to providing a holistic, cost-effective, and appropriate approach to CCTS systems and respective integrity management. This involves bringing together a diverse range of engineering disciplines including geology, reservoir engineering, processing, pipeline management, materials selection, and corrosion. Such an integrated approach will have a major impact on facilities, product optimization, health, security, and the environment, ensuring continuous, safe, and effective operations.
References
1 A. Dugstad, M. Halseid, B. Morland, Clausen, “Experimental Techniques Used for Corrosion Testing in Dense Phase CO2 with Flue Gas Impurities,” CORROSION 2014 paper no. 4383 (Houston, TX: NACE International, 2014).
2 S. Papavinasam, Corrosion Control in the Oil and Gas Industry (New York, NY: Elsevier, 2013).
3 “Energy Technology Perspectives 2010, Scenarios & Strategies to 2050” (Paris, France: International Energy Agency, 2010).
4 Y. Xiang, Z. Wang, M. Xu, Z. Li, W. Ni, “A Mechanistic Model for Pipeline Steel Corrosion in Supercritical CO2-SO2-O2-H2O Environments,” J. of Supercritical Fluids 82 (2013): pp. 1-12.
5 ASME B31.8, “Gas Transmission and Distribution Piping Systems” (New York, NY: ASME).
6 E. De Visser, C. Hendriks, M. Barrio, M.J. Mølnvik, G. de Koeijer, S. Liljemark, Y.L. Gallo, ”Dynamis CO2 Quality Recommendations,” Int. J. Greenhouse Gas Control 2 (2008): pp. 478-484.
7 “IPCC Special Report on Carbon Dioxide Capture and Storage,” prepared by Working Group III of the Intergovernmental Panel on Climate Change (Cambridge, United Kingdom: Cambridge University Press, 2005), p. 442
8 K. Havens, Kinder Morgan presentation at the Indian Center for Coal Technology Research.
9 NETL Quality Guidelines for Energy System Studies, ”CO2 Impurity Design Parameters,” DOE/NETL-341/011212, 2012.
10 J.M. West, ”Design and Operation of a Supercritical CO2 Pipeline-Compression System Sacroc Unit, Scurry County, Texas,” SPE paper no. 4804 (1974).
11 T. Maldal, I.M. Tappel, “CO2 Underground Storage for Snøhvit Gas Field Development,” Energy 29 (2004), p. 1403.
A version of this article was published in the March 2014 issue of Pipeline & Gas Journal.